KFU scientists describe new mechanisms of hydrate formation with various surfactants
Gas hydrates look like ordinary snow or ice, but they burn readily. This is due to gas molecules securely “packed” within a crystal lattice composed of water molecules. These compounds form at high pressure and low temperature and have great potential as containers for the safe storage and transportation of methane, hydrogen, and carbon dioxide. They are environmentally friendly and stably stored at temperatures of minus 15 to minus 20 degrees Celsius and atmospheric pressure.
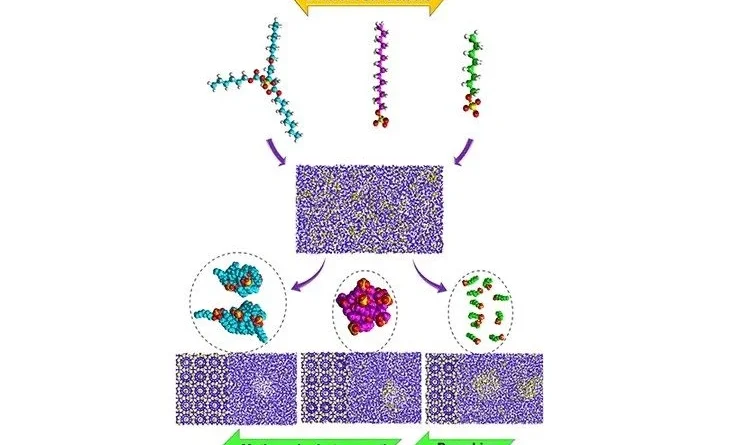
The paper in ACS Applied Materials & Interfaces how to accelerate hydrate formation and increase gas absorption to create efficient energy storage systems. The study was conducted by the Research Laboratory of Hydrate Technologies for Greenhouse Gas Utilization and Storage with the support of the Ministry of Science and Higher Education of the Russian Federation. “We have described in detail the molecular mechanisms of gas hydrate formation using a wide range of surfactants differing in chemical structure, charge, and hydrophobic-hydrophilic balance,” explains senior researcher Abdolreza Farhadian. “To establish the mechanisms of interaction between surfactant molecules, water, and gases such as methane, we used molecular dynamics methods in combination with experimental observations. Ultimately, we identified two main mechanisms facilitating hydrate formation: adsorption at the phase interface (gas-liquid or hydrate-liquid) and the formation of micelles or aggregates within the bulk of the solution.”
The experiments involved 16 surfactants from various groups – cationic, anionic, and nonionic – that differ in structure, polar head type, hydrocarbon chain length, and degree of hydrophobicity.
“We have established clear relationships between molecular structures and their impact on hydrate formation, which had previously been insufficiently studied. Furthermore, we were able to demonstrate that even small changes in molecular structure can significantly affect hydrate growth kinetics and the degree of methane absorption,” continues the scientist. “For example, surfactants with long hydrophobic chains (more than 12 carbon atoms) significantly accelerate hydrate formation due to stronger interactions with gas molecules. It was found that the presence of aromatic groups promotes accelerated hydrate growth but can lead to increased foaming during hydrate decomposition. All this points to the need to consider several factors when developing effective surfactants.”
According to Farhadian, the team’s identification of competing molecular interactions that determine the hydrate formation process is of great scientific significance.
“On the one hand, hydrophobic interactions between the non-polar tails of the surfactant and gas molecules promote gas accumulation near the hydrate surface, accelerating its formation. On the other hand, hydrogen bonds between the polar heads of surfactants and water molecules can disrupt the hydrogen bond structure necessary for hydrate crystal lattice formation, thereby slowing the process. This dual effect leads to a more complex understanding of the role of surfactants, showing that they can both accelerate and inhibit hydrate formation depending on their properties and environmental conditions,” he says.
Another important aspect is considering the balance between the rate of hydrate formation and the rate and efficiency of gas extraction.
“Foam formation during hydrate decomposition can hinder gas extraction and reduce system efficiency,” notes Abdolreza Farhadian. “Branched surfactants, for example, are promising for practical use; they exhibit a high rate of hydrate formation and reduced foaming during decomposition,” he said.
The article provides specific recommendations for the selection and design of surfactants for various applications.
“The established relationships between the structural characteristics and efficiency of surfactants enable the targeted development of substances with desired properties. This is especially important for industrial applications, where it is necessary to consider not only efficiency but also cost effectiveness, stability, and performance characteristics. Surfactant design opens up opportunities for the creation of new technologies in clean energy and environmental protection,” the researcher states.
According to Farhadian, the new scientific results obtained by the international research team will help accelerate the implementation of gas hydrate technologies.
“The development of chemical reagents that increase the rate of hydrate formation and gas conversion to hydrate form, as well as methods for improving the stability of the resulting pellets and the development of regasification methods are the main objectives of the hydrate technologies laboratory. We conduct both fundamental research to identify the mechanisms of action of various hydrate promoters and inhibitors, as well as applied research in collaboration with industrial partners for specific industrial applications,” comments lab supervisor Mikhail Varfolomeev.