IGPT scientists continue research of promoters for enhanced methane hydrate formation
The new paper describes promoters of hydrate formation based on amino acids and gluconic acid.
Shamil Gainullin, a junior research associate at the Laboratory of Hydrate Technologies for Greenhouse Gas Utilization and Storage, discussed the advantages of storing and transporting natural gas as gas hydrates and a new “growth stimulator” developed at KFU for these solid, crystalline compounds, which resemble ice.
“Natural gas remains a key energy source, but its storage and transportation still pose significant technical challenges. Typically, this involves either liquefying the gas at very low temperatures or compressing it at high pressure. Both methods require complex infrastructure, significant energy consumption, and stringent safety requirements. One of the safest ways to store methane is in the form of gas hydrates, where it is stored in a bound state,” explained the young scientist.
According to the researcher, hydrate technology, as an alternative to traditional methods of storing and transporting natural gas, can be used in areas where pipeline construction or handling liquefied natural gas is economically impractical.
Representatives of the laboratory have been working to improve the efficiency of this technology for several years.
“The main obstacle we face is slow kinetics. Hydrate begins to form late, either grows slowly, or the water does not completely convert to hydrate,” explained Shamil Gainullin. “Furthermore, a dense hydrate film can form at the gas-water interface, which prevents further phase contact and further slows the process.”
Various promoters, most commonly surfactants, are used to accelerate hydrate formation, but they have a number of drawbacks.
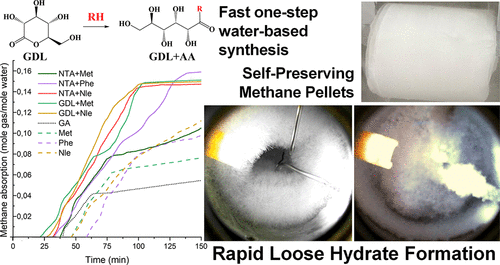
“Amino acids can accelerate methane hydrate formation. They are biodegradable, non-toxic, and do not promote foaming like traditional surfactants. Unfortunately, these substances are inferior to classic industrial promoters,” continued the interviewee. “We chemically modified amino acids to improve their effectiveness and studied new compounds synthesized based on amino acids and gluconic acid.”
He noted that the natural, biocompatible compound gluconic acid was chosen as a modifier, firstly, because of its environmental friendliness, and secondly, because it contains many hydroxyl groups capable of forming hydrogen bonds and influencing the organization of water molecules, which, in turn, can accelerate the onset of hydrate formation.
“An important aspect is the simplicity of synthesis,” noted the researcher. “The starting reagent is glucono-delta-lactone. This readily available food additive, E575, enables single-step synthesis in water, under mild conditions and without purification steps, and allows for the production of promoter solutions directly at the point of use. Complex chemical equipment is not required.”
The effect of synthesized biopromoters on methane hydrate formation was studied in high-pressure autoclaves under static and dynamic conditions, as well as using differential scanning calorimetry, visual observation, granulation, stability testing, molecular dynamics, and quantum chemical modeling.
The results of the study are presented in a paper published in the journal Energy & Fuels.
“The main conclusion is that the new group of biopromoters significantly accelerated methane hydrate formation even at very low concentrations: the component mass percentage in the mixture was 0.05. In experiments, hydrate formation in the presence of these compounds initiated in approximately 19-25 minutes, compared to 54 minutes in the presence of a well-known surfactant such as SDS, and approximately 45 minutes for unmodified amino acids. In other words, the new compounds acted more quickly and initiated the process earlier. At the same time, they not only accelerated the start-up but also ensured high hydrate yields: under dynamic conditions, the conversion of water to hydrate reached 88-96 percent, and methane absorption reached 0.160 moles per mole of water,” Gainullin concluded.
The study also found that the resulting hydrate pellets possessed high density, mechanical strength, and stability during methane storage, the researcher emphasized.
“Hydrates obtained in the presence of conventional surfactants foam heavily during gas release, making the technology less convenient for industrial use. In the systems we created based on amino acids and gluconic acid, this effect was not observed either during hydrate formation or during its decomposition,” he noted.
The competitive advantages of the obtained compounds are: natural origin, simplicity and low cost of their synthesis, high hydrophilicity, and good kinetic characteristics.